What is National Donate Life Month? National Donate Life Month, observed each April, helps raise awareness about donation; encourages Americans to register as organ, eye, and tissue donors; and honors those who have saved lives through the gift of donation. For some people with stage IV colorectal cancer who have unresectable metastases to the liver, a liver transplant may be their opportunity to become cancer free.
Celebrating Living Donors Who Gave People with Stage IV Colon Cancer a Second Chance at Life
Currently, living donor liver transplant surgery can be done for people with stage IV colon cancer or stage IV rectal cancer with metastases to the liver.
However, if the colorectal cancer has metastasized to areas other than the liver, a person will not be a candidate for the surgery. “One out of 10 patients with stage IV colorectal cancer with unresectable liver metastases will fill the criteria for liver transplant surgery,” according to Roberto Hernandez-Alejandro, MD, FACS, FRCSC, Professor of Surgery; Chief, Division of Transplantation, Hepatobiliary Surgery, University of Rochester.
Once the candidates are determined to be eligible for liver transplant surgery, they become charged with finding their own liver donors.
“Colorectal cancer patients don’t have liver disease. They have cancer in the liver. Their liver function is normal,” said Dr. Hernandez. The people who are most ill with liver disease, such as cirrhosis or hepatitis, are at the top of the list to receive donated livers. But because people who have stage IV colorectal cancer who are candidates for liver transplant surgery have a liver that is considered “healthy,” they would fall to the bottom of the list to receive a donated liver.
Here is the power of living donors liver transplant surgeries: Not only do living donors save lives, but they also have the ability to select the person to whom they will donate their liver, which is exactly what happened with both Carole Motycka-Mancini, a stage IV colon cancer survivor who received a living donor liver transplant five years ago, and Tim McDonald, a stage IV colon cancer survivor who will receive a living donor liver transplant on May 16, after waiting almost 15 months for a donor.
Carole and Tim’s stories are celebrating living donors during National Donate Life Month, as well as highlighting the need and importance of living donors.
Carole’s Story
Carole was 42 years old and had just finished a 17-mile hike when she ended up in the emergency room with shoulder pain and was diagnosed with stage IV colorectal cancer in April 2016.
After 12 rounds of chemo, a colon resection, and a three-stage liver resection where surgeons removed the right lobe and took tumors off the left lobe (including a complex tumor on the hepatic flexure), Carole was declared no evidence of disease (NED). At this point, Carole was in “clean-up mode” with an HAI (Hepatic Artery Infusion) pump, which delivered intensive chemo directly to her liver.
Carole’s HAI pump was in place to help tumors shrink and necessary to help her maintain NED status. But in July 2017, six pump treatments in, the chemotherapy that saved Carole’s life from stage IV colon cancer caused her liver to begin to fail.
Multiple surgeries with drains in an attempt to heal her liver yielded no progress. For months her medical team tried to control her liver biliary issues, resulting in a whopping 19 surgeries to place drains and stents. Ultimately, Carole had a liver aneurysm in the parking lot of Cleveland Clinic and was rushed into surgery.
That’s when Carole’s liver doctor, Frederico Aucejo, MD, the Director of the Liver Cancer Program and Surgical Director of the Liver Tumor Clinic in the Department of General Surgery at Cleveland Clinic advised Carole that he had exhausted almost all efforts to save her life – except one: living donor liver transplant surgery.
Pioneered in Norway, the living donor liver transplant surgery was a relatively new procedure at the time in the U.S. But Carole, the mother of four boys, knew exactly what she had to do.
I wanted to make sure my kids knew that I never gave up on them.
-Carole Motycka-Mancini, stage IV survivor and liver recipient
Without the transplant, Carole likely would die. But the liver transplant – even with the risks involved – would give Carole another chance.
I said ‘Yes.’ It was like a light in the dark.
-Carole Motycka-Mancini
Finding Her Donor
Carole needed to find her own donor, and she asked her surgeon how to go about finding one. “Facebook Marketplace” was not the solution. But her church bulletin was.
At the time, Carole was the Youth Director at her church. Her pastor placed a request to help Carole in the church bulletin asking anyone who fell within certain parameters of eligibility to donate and interested in donating a portion of their liver to contact Carole’s transplant team at the Cleveland Clinic. Fifty people responded. While Carole didn’t know her liver donor personally, she knew of him from her church.
Five years ago, on April 23, 2018, Carole received her new liver.
Responding to a Request and a Call
Jason Stechschulte, a member of Carole’s church, knew immediately when he saw the request for help for Carole in the church bulletin that he was going to be Carole’s donor match.
“There’s really no other way to explain it, other than the Holy Spirit was enveloping me. I just knew I was going to be the donor,” said Jason.
Jason was the sixth person screened, and the person who would become Carole’s liver donor.
Being a donor isn’t just one person donating: The entire family is affected. “There’s a lot of commitment to being the spouse to a liver donor,” said Stephanie, Jason’s wife. She admitted to having concerns before the surgery.
I was afraid that Carole would die before she got her liver. I was afraid that something would happen to Jason ranging from long-term health issues to death. I was worried that I would be like a single mom for many weeks, and I didn’t know if I would be good at it.
–Stephanie Stechschulte, wife and caregiver of liver donor Jason Stechschulte
In addition to taking care of Jason as he recovered, Stephanie also had to take care of their two children and household. Fortunately, their church members, friends, and family graciously stepped in to assist and get them through this period.
Jason said the surgery was easy, and he only remembers pain on the third or fourth night, and only for one night. Otherwise, his pain was easily managed with Tylenol® or Advil®. After his incision healed, Jason felt no different, having just donated a hefty portion of his liver. When he returned to work, people were shocked that he looked so good for having been through such a grueling surgery.
The most rewarding part of this experience is also the most humbling. Knowing God asked me to play a part in this and then seeing everyone else pitch in to contribute and make it happen. I had the easy part, since I was able to just sleep through it and get a mini-vacation. Everyone else went out of their way to make this possible.
–Jason Stechschulte, liver donor
Jason is astounded that Carole’s life was saved with his liver and an amazing team of doctors, while he was sleeping in the next room over.
Carole’s transplant team at the Cleveland Clinic explained the process thoroughly to Jason, making sure he understood the risks and was as comfortable as possible through the process. Jason “absolutely recommends” being a living donor for liver transplant surgery.
Frederico Aucejo, MD, on Carole’s Liver Transplant Surgery
 In April 2018, Frederico Aucejo, MD, the Director of the Liver Cancer Program and Surgical Director of the Liver Tumor Clinic in the Department of General Surgery at Cleveland Clinic, performed one of the first stage IV colorectal cancer liver transplant surgeries in the United States on Carole. According to Dr. Aucejo, “Carole’s case was an example of the utilization of all treatments available today in the toolbox for colorectal cancer liver metastases.”
In April 2018, Frederico Aucejo, MD, the Director of the Liver Cancer Program and Surgical Director of the Liver Tumor Clinic in the Department of General Surgery at Cleveland Clinic, performed one of the first stage IV colorectal cancer liver transplant surgeries in the United States on Carole. According to Dr. Aucejo, “Carole’s case was an example of the utilization of all treatments available today in the toolbox for colorectal cancer liver metastases.”
“Her case is particularly compelling considering the burden of disease her liver had when she was diagnosed, and the multiple interventions she received including systemic and liver directed chemotherapies, advanced liver resections, and liver transplantation with a live donor. People should know that even in cases so advanced a good outcome like Carole’s is possible as she presents no evidence of tumor recurrence at five years post-transplant. At the same time, people should know that successful stories like this are infrequent; and therefore, there is much work to do by the scientific community to continue improving patient selection, efficient drug discovery and better tumor biology understanding,” said Dr. Aucejo.
Carole’s Five Year Anniversary of her Live Donor Liver Transplant Surgery
This month, National Donate Life Month, Carole celebrates her five year anniversary of her live donor liver transplant surgery. She continues to receive scans and blood work every four months. She also takes immunosuppression medicine — as any person who received a transplant would — as well as self-administers Lovenox® shots for some clotting issues, and she practices overall good health habits.
We do a good job getting the word out and discussing this in Colontown, a partner of Fight CRC. I am an administrator of ‘Transplantation Station,’ a place where we discuss all-things transplant. It’s important to remember it’s a small subset of patients that qualify at this time, and the opportunity is complex.
–Carole Motycka-Mancini
Tim’s Story
Tim McDonald was diagnosed with stage IV colon cancer with metastases to his liver in November 2020. One oncologist told Tim that he had three years to live. Another oncologist told Tim his life expectancy would be “in the five-year range.”
When Tim went to Moffitt Cancer Center in Tampa, his doctor didn’t discuss his life expectancy, but rather how they could fight Tim’s cancer.
Initially told he would be on chemo for life and was not a suitable candidate for liver resection, Tim learned about Colontown through Man Up to Cancer and was put in touch with Dr. Hernandez who performs liver transplants at University of Rochester Strong Memorial Hospital.
In March 2022, Tim had his donor meeting, and he believed obtaining a live donor for his stage IV colorectal cancer living donor liver transplant surgery would be the relatively easy part. Fifteen months later, Tim said finding a liver donor was more difficult and frustrating than he thought it would be.
On April 3, 2023, while celebrating his wife’s birthday on a cruise, Tim learned that he now has a liver donor, and he will be undergoing stage IV colorectal cancer liver transplant surgery on May 16, 2023.
More than a week after learning Tim’s transplant team found a donor for him, April 12 was another big day for Tim when he rang the bell signaling the end of chemo treatment, something he never expected he would get the opportunity to do.
Preparing for Liver Transplant Surgery
Tim’s story is a little different from Carole’s since he was responding well to treatment without the devastating effects that Carole’s treatment caused to her liver.
With a colon resection surgery completed in March 2022 and other concerns addressed, Tim was relieved to receive the results that everything was OK, and he could continue on toward liver transplant surgery. His donor search officially began.
Although an optimistic and patient person by nature, Tim was surprised that the search for a liver donor took longer than he anticipated. Out of everyone he knew who had liver transplant surgery, finding his donor took the longest.
When he received news of his liver donor and was given a living donor liver transplant surgery date, Tim learned that he needed to be off chemotherapy for four to six weeks, so his body could recover from surgery more quickly. His final chemo treatment placed him firmly in that four to six weeks window, and Tim was able to ring that bell, something he never thought he would be able to do since he believed he was on chemo for life.
Tim’s pre-op visit is scheduled a week before his surgery. He will get a chest x-ray, a CT scan, and more blood work. All of his other screening had been done during a two-day evaluation in December 2022, when Tim’s transplant team initially believed they had found Tim’s donor. At that time, he had the stress echo test, and the initial meetings with everyone working on him and his care team. They took 27 vials of blood, the most blood Tim said he has ever had taken.
Tim is prepared. He has been eating healthy, not drinking or smoking. He feels he has prepared his body to go through surgery most effectively and to recover quickly. Finally, he knows how important it is to walk and move post-surgery.
Potential Donor Disappointments
For stage IV colorectal cancer live donor liver transplant surgery, candidates need to get their own donor (directed donor), where the donor needs to call the transplant team at the facility the recipient is being treated, give the name of the person who needs the liver, and date of birth.
During his donor search, Tim specifically did not ask anyone he knew – not friends, family, or even acquaintances – for a liver donation because he didn’t want them to feel obligated or pressured.
Over the past 15 months, Tim met or had people reach out to him who attempted to become donors for him. His research revealed that most people average five to seven potential donors before they found their donor match. There were at least nine people – but probably 10 – that Tim knew of who were screened as a potential donor beyond the initial phone call before he found his match.
To paraphrase Mike Tyson, “Everybody’s got a plan until they get punched in the face.”
I think getting cancer is getting punched in the face. I think going through this donor process and not knowing that a potential match – why they weren’t, and why they’re not anymore – is a punch in the face. And every time you get that, you need to figure out how you can react to it.
–Tim McDonald, stage IV survivor and liver recipient scheduled for live donor liver transplant surgery on May 16
Ultimately Tim realized the only thing he can control is his reaction, not the situation. He no longer focuses on the control he thought he had, but instead focuses on his reaction to the news he is getting.
So although he has received disappointing news while waiting for his liver donor to be found, Tim no longer felt depressed or resentment. He kept moving forward.
The Silver Lining
While he was patiently awaiting his own liver donor, Tim realized there was a need for people who may not be donor matches for him, but who could help others. He created a site, Share My Liver, in an effort to help guide others who have stage IV colorectal cancer with unresectable metastases to the liver to find their donors.
The same week Tim found out that his transplant team had located a liver donor for him, the first confirmed match on Share My Liver came through. Tim is overjoyed.
Meeting His Donor and His Happily Ever After
Tim had an hour-long conversation with his live donor, and he is looking forward to meeting her and her husband for dinner in person when he travels to Rochester, New York, for his surgery.
What Tim is most excited about with his liver transplant surgery is that this surgery provides the highest projected rate of long-term survival compared with any other treatment.
Roberto Hernandez-Alejandro, MD, FACS, FRCSC, on Tim and Living Donor Liver Transplants
 In May, Roberto Hernandez-Alejandro, MD, FACS, FRCSC, Professor of Surgery; Chief, Division of Transplantation, Hepatobiliary Surgery, University of Rochester, will perform Tim’s live donor liver transplant surgery at Strong Memorial Hospital, Rochester, New York. Dr. Hernandez is looking forward to helping Tim become cancer free. According to Dr. Hernandez, “Liver transplantation is for a specific and a very narrow population who has stage IV colon cancer. Tim has fallen into this small population of people eligible for a liver transplant from a living donor. Imagine having a 30% chance of being cured instead of surviving to five years with a 5% to 10% chance. With liver transplantation, there is a 50% chance of surviving at 10 years. This is a huge difference and also factors in the quality of life of liver transplantation as well.”
In May, Roberto Hernandez-Alejandro, MD, FACS, FRCSC, Professor of Surgery; Chief, Division of Transplantation, Hepatobiliary Surgery, University of Rochester, will perform Tim’s live donor liver transplant surgery at Strong Memorial Hospital, Rochester, New York. Dr. Hernandez is looking forward to helping Tim become cancer free. According to Dr. Hernandez, “Liver transplantation is for a specific and a very narrow population who has stage IV colon cancer. Tim has fallen into this small population of people eligible for a liver transplant from a living donor. Imagine having a 30% chance of being cured instead of surviving to five years with a 5% to 10% chance. With liver transplantation, there is a 50% chance of surviving at 10 years. This is a huge difference and also factors in the quality of life of liver transplantation as well.”
The Screening Process for Donating a Liver
When someone decides to donate a liver, it’s not as simple as a phone call or email that puts them on a list.
Tim’s experience is that screening is a two- or three-month-long process per potential eligible donor. The screening process entails:
- An initial phone call
- An evaluation phone call
- Blood work
- Sending medical records
- Colonoscopy
- CT scan
- Liver ultrasound
This screening process occurs one person at a time and one step at a time. There are never multiple people being screened to become a donor for the same recipient. While the process is methodical, it is also stressful to the recipient who is waiting to learn of a match.
Interesting Facts About Stage IV Colorectal Cancer Liver Transplant Surgery
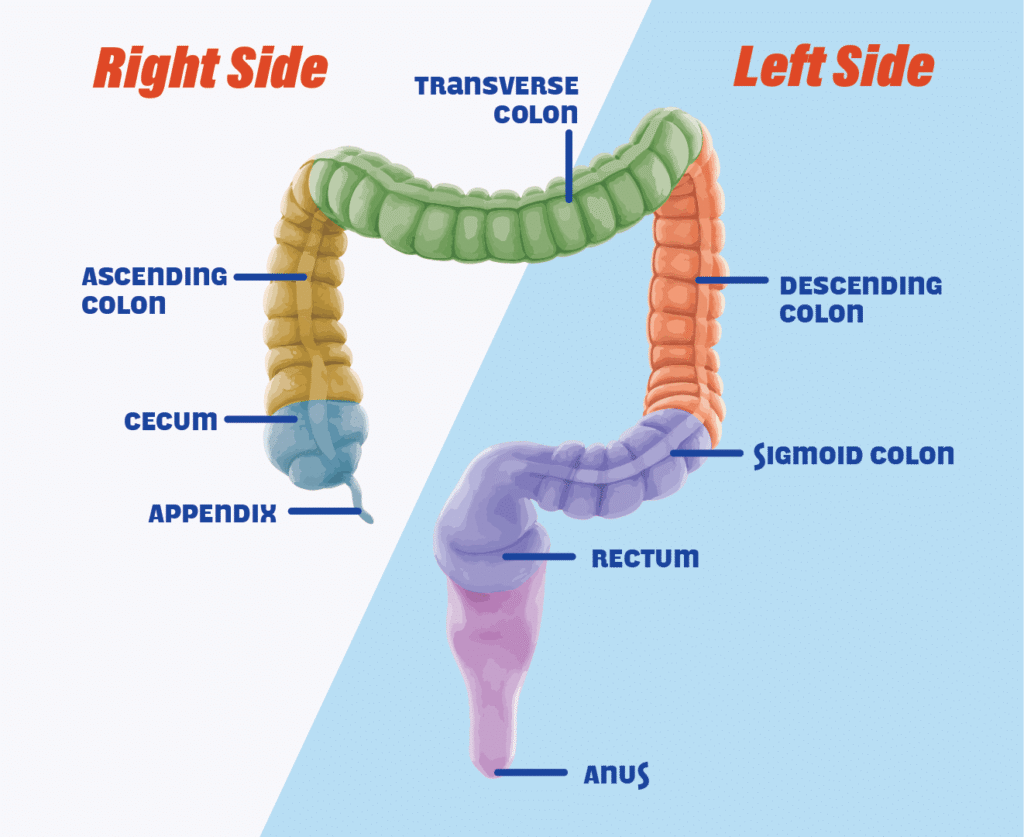
Right Side vs. Left Side
So far, Dr. Hernandez says that his team has done fewer right-side colon cancer live donor liver transplant surgeries compared with left-side colorectal cancer transplant surgeries. Right-side colon cancer is typically more aggressive, so the liver transplant team needs to be more cautious with people who have colon cancer on the right side.
Liver Transplant Surgery in the United States
Liver transplant surgery for people with stage IV colorectal cancer has only been performed in the United States for the past five years. To date, more than 40 live donor liver transplant surgeries have taken place.
We need to educate people that this is a possibility that exists. Physicians and patients may think a liver transplant is the last resort. If a patient is diagnosed with stage IV colorectal cancer with metastases only to the liver, then we can meet and generate a plan. First the patient will be treated with chemo. Next the primary tumor will be removed. Then we can begin the work of setting up the process for living donor transplantation.
–Dr. Roberto Hernandez
As someone who had all but run out of options, Carole recommends consulting your physician team to find what fits best for you and the opportunities that exist for your particular cancer profile.
Five years ago, it was just me. Today, there are more than 40 of us who have transplanted.
–Carole Motycka-Mancini
People who donate their livers go on to live a completely normal life. Their liver regenerates. If you donate the right side of your liver, you don’t grow a new right side, but the left side grows to compensate for the portion missing from the right side. The liver can regenerate anywhere from 80% to 105%, according to Dr. Hernandez.
Dr. Hernandez’s final suggestion is not to lose hope. If a person is told they are not a candidate for stage IV colorectal cancer liver resection surgery, they should not hesitate to seek out a second or third medical opinion if they feel they need it. Dr. Hernandez has seen people who were told they had unresectable tumors in their liver, but when he reviewed imaging, he saw these tumors could be resected.
To Learn More
Many organizations accept organ or tissue donations. If you’re interested in donating for someone specific such as Tim through the University of Rochester. Medical Center Liver Transplant Program, or Carole through the Cleveland Clinic Transplant Team, be sure to contact the potential recipient’s hospital’s transplant team for more information and instruction on how to proceed and get screened to give a second chance at life to people with stage IV colorectal cancer.


 In April 2018, Frederico Aucejo, MD, the Director of the Liver Cancer Program and Surgical Director of the Liver Tumor Clinic in the Department of General Surgery at Cleveland Clinic, performed one of the first stage IV colorectal cancer liver transplant surgeries in the United States on Carole. According to Dr. Aucejo, “Carole’s case was an example of the utilization of all treatments available today in the toolbox for colorectal cancer liver metastases.”
In April 2018, Frederico Aucejo, MD, the Director of the Liver Cancer Program and Surgical Director of the Liver Tumor Clinic in the Department of General Surgery at Cleveland Clinic, performed one of the first stage IV colorectal cancer liver transplant surgeries in the United States on Carole. According to Dr. Aucejo, “Carole’s case was an example of the utilization of all treatments available today in the toolbox for colorectal cancer liver metastases.” In May, Roberto Hernandez-Alejandro, MD, FACS, FRCSC, Professor of Surgery; Chief, Division of Transplantation, Hepatobiliary Surgery, University of Rochester, will perform Tim’s live donor liver transplant surgery at Strong Memorial Hospital, Rochester, New York. Dr. Hernandez is looking forward to helping Tim become cancer free. According to Dr. Hernandez, “Liver transplantation is for a specific and a very narrow population who has stage IV colon cancer. Tim has fallen into this small population of people eligible for a liver transplant from a living donor. Imagine having a 30% chance of being cured instead of surviving to five years with a 5% to 10% chance. With liver transplantation, there is a 50% chance of surviving at 10 years. This is a huge difference and also factors in the quality of life of liver transplantation as well.”
In May, Roberto Hernandez-Alejandro, MD, FACS, FRCSC, Professor of Surgery; Chief, Division of Transplantation, Hepatobiliary Surgery, University of Rochester, will perform Tim’s live donor liver transplant surgery at Strong Memorial Hospital, Rochester, New York. Dr. Hernandez is looking forward to helping Tim become cancer free. According to Dr. Hernandez, “Liver transplantation is for a specific and a very narrow population who has stage IV colon cancer. Tim has fallen into this small population of people eligible for a liver transplant from a living donor. Imagine having a 30% chance of being cured instead of surviving to five years with a 5% to 10% chance. With liver transplantation, there is a 50% chance of surviving at 10 years. This is a huge difference and also factors in the quality of life of liver transplantation as well.”